Facts You Might Want to Have Known Before You Accepted the COVID-19 mRNA Vaccine: Part 1
These are serious issues that go right to the heart of the matter: Who owns your body, you, or the State? What did FDA and CDC know and when, and why have they not issued more warnings?
If you visit the CDC website in search of known adverse events following the COVID-19 mRNA shots, you’ll find a page that lists a few, but that quickly assures you that the risk is low. The page tells you that individuals have experienced anaphylaxis, thrombosis with thrombocytopenia syndrome, aka, immune thrombocytopenia, myocarditis or pericarditis, and Guillain-Barré Syndrome (GBS), and that the risk of each of these is “low”.
The page does not define “low”, and in keeping with best practices of accountability to the public fails to quantify the risk in terms that are understandable to everyday people.
These issues are par for the course of the CDC’s reporting on vaccine adverse event risk. Most often, however, resources eschew the word “risk” altogether, using euphemisms such as “Safety Signal”.
I recently cataloged the number of ways the CDC, FDA, and others failed to inform the public about risks that were known - that is, they already knew about certain risks and proceeded with putting policies into place after delaying the release of key information that may have influenced individuals’ and organizations’ willingness to go along with the COVID-19 mass vaccination effort.
This is information that CDC and FDA should be making waves about - and yet they remain silent.
The first of these is new onset autoimmunity, which is diverse in form and not at all rare. I review this in detail in the article Immunosuppression Wars 1.0. Why are the FDA and CDC silent and failing to issue warnings when the scientific literature is filled with studies and reports characterizing autoimmune conditions triggered by COVID-19 vaccines?
FDA did issue a warning about heart inflammation on June 26, 2021, but why? Three days earlier, Dr. Tom Shimabukuro told the VRBAC Committee that after analyzing millions of records in the Vaccine Safety Datalink he could not find any signal trigger for myocarditis or pericarditis. VRBAC voted under the influence of Shimabukuro’s result; of course, they approved of the vaccine.
What if an independent study could have been conducted to determine if the VAERS database contained a safety warning that might have informed the VRBAC committee - or the ACIP committee - that myocarditis was a potential risk prior to their vote?
Oh, that’s right, there was. My colleagues Jessica Rose and Peter McCullough had their study accepted, then “unaccepted”, from an Elsevier journal over a difference in interpretation.
This study, which found an increased risk in teens, would have provided a baseline for future studies following boosters.
The journal, “Current Problems in Cardiology”, pulled the article after accepting it following no peer review, with the following explanation: “The Publisher regrets that this article has been temporarily removed. A replacement will appear as soon as possible in which the reason for the removal of the article will be specified, or the article will be reinstated.”
Eventually, the journal reported “This article has been withdrawn at the request of the author(s) and/or editor. The Publisher apologizes for any inconvenience this may cause.”
This doublespeak is misleading; Jessica and Peter did not request the withdrawal of the study, so why write it this way?
Genome Modification and Potential Genomic Instability
Three early studies showed that contrary to reassurances made by naysayers, the mRNA from COVID-19 mRNA jabs could, in fact, become inserted following reverse transcription into human cells.
A more recent report found that at least 10-20% of people injected with mRNA vaccines integrated the mRNA in whole or part into their genome. The study, mentioned in this review was published in September of 2021 in Virus Research:
“Motivated by reports about prolonged shedding of SARS-CoV-2 RNA and continued positive RT-PCR tests among survivors of Covid-19, Zhang et al. investigated whether SARS-CoV-2 RNA could be reverse transcribed into DNA that in turn might integrate into the host genome and continue to be transcribed into RNA (Zhang et al., 2021). By nucleotide sequence analyses, the authors observed SARS-CoV-2 DNA copies in the genomes of about 10 to 20% of Covid-19 patients. This SARS-CoV-2 DNA was flanked by target site duplications and consensus LINE-1 endonuclease recognition sequences. The latter two characteristics were not always observed. Thus LINE-1-independent mechanisms for SARS-CoV-2 DNA integration were also conceivable. It was emphasized that only sub-genomic parts of SARS-CoV-2 DNA were found integrated. This type of integration precludes the possibility of infectious viruses being produced by those who carry fragmented viral SARS-CoV-2 DNA. The integrated parts of SARS-CoV-2 DNA were frequently transcribed. The authors concluded that SARS-CoV-2 infection could lead to the synthesis of endogenous LINE-1 retrotransposon-encoded reverse transcriptases. These enzymes generated DNA retro-transcripts. Possibly only fragments of this DNA were inserted into the human genomes and transcribed into SARS-CoV-2 RNA. This finding might have implications in the context of long-term sequelae of SARS-CoV-2 infections for the genomes of Covid-19 patients.”
The study was published in the US’s Proceedings of the National Academy of Sciences (PNAS; (See "Reverse-transcribed SARS-CoV-2 RNA can integrate into the genome of cultured human cells and can be expressed in patient-derived tissues".
Despite this knowledge, there have been no studies on the carcinogenicity. The US FDA still distributes the vaccine product insert with the following language:
A September 2020 “confidential” appendix to the clinical trial studies submitted for the Pfizer/BioNTech COVID vaccine for EU approval, titled “Justification for the absence of studies in CTD Module 4” notes under “Safety Pharmacology” that “No safety pharmacology studies were conducted as they are not considered necessary according to the WHO guideline.”
"Carcinogenicity testing is generally not considered necessary to support the development and licensure of vaccine products for infectious diseases," Pfizer reported.
Jessica Rose did a good write-up on this a year ago.
The consequences of the US FDA and CDC failing to warn about the insertional mutagenesis are profound and can include an increased risk of cancer, and of course, genome integration can lead to autologous spike protein production, leading to (among other things) autologous spike protein production and immune tolerance. Immune tolerance means that the vaccinated person’s immune system may no longer be able to distinguish a non-self-spike protein from a viral spike, with potential consequences for keeping up with new variants.
Teens with myocarditis have far more freely circulating spike protein than could be expected due to the vaccine dose. In a study published in January 2023 in the American Heart Association’s journal Circulation, the paper Circulating Spike Protein Detected in Post–COVID-19 mRNA Vaccine Myocarditis, the authors studied 16 myocarditis victims and 45 unaffected individuals, all of which were aged between 12 and 21. Each had accepted either the Moderna mRNA-1273 or the Pfizer-BioNTech BNT162b2 injections. The vaccinated patients had far more copies of the spike protein than could be explained by the number of mRNAs delivered to cells from the injection, meaning autologous transcription of spike mRNA from the human genome, where it should not be found.
Blood Clots Buried via Statistical Shamwizardry
This study by Peter Elkin et al., which specifically addresses concerns over clotting claims to find no association between COVID-19 mRNA jabs and clotting. Their results, however, are dependent on an “adjustment” using “age, sex, BMI, 2-year Elixhauser score, and race…”
The Elixhauser score is a multi-component “comorbidity” index (causal relationships assumed) that includes… clotting disorders.
The authors concluded:
“The results provide reassurance that there is only a trivial increased risk of VTE with the current US SARS-CoV-2 vaccines used in veterans older than age 45. This risk is significantly less than VTE risk among hospitalized COVID-19 patients. The risk-benefit ratio favors vaccination, given the VTE rate, mortality, and morbidity associated with COVID-19 infection.”
But adjusting for the outcome variable this means the study adjusted for the outcome as a confounder. It’s a bogus study.
But that did not stop the University of Buffalo from issuing this truly embarrassing press release:
In the press release, the lead author quipped “We wanted to know the truth”. Well, the truth is you cannot adjust for the dependent variable and expect to find the truth.
The question is not “How could these issues exist when Pfizer, Moderna, FDA, and CDC all assured us that mRNA injection technologies were safe?”
The question is “What did Pfizer and Moderna know, what did FDA and CDC know, and why did they not inform the public?
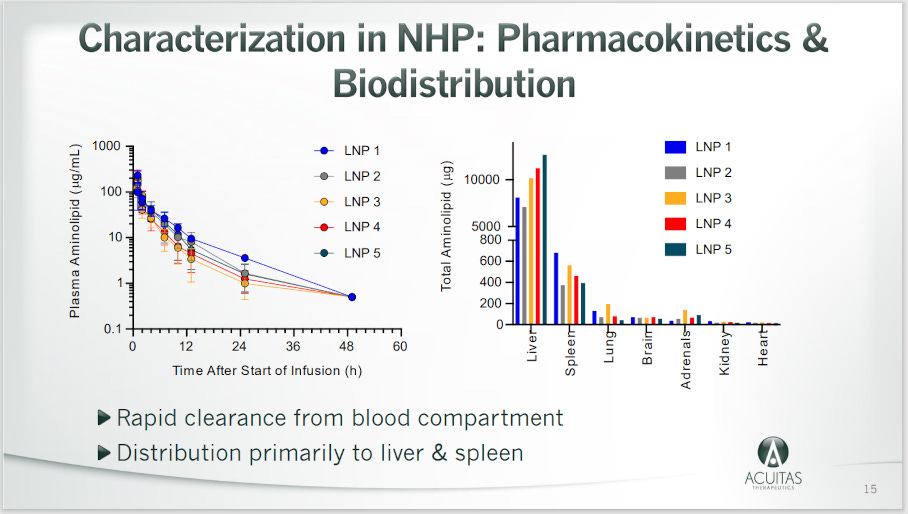
They Knew the mRNA LNPs Spread Throughout the Body
The Bahl et al. biodistribution study, “Preclinical and clinical demonstration of immunogenicity my mRNA vaccines against h10N8 and h7n9 influenza vaccines” is proof they knew that shots of this type would not stay in the muscle. Here’s their Table 1. The date? April 2017.
Acuitas, a company tasked to study the mRNA shot distribution in mice, found the same issue, reported in a “non-confidential” presentation available online:
The date? May 17, 2021.
mRNA-Containing LNPs Can be Found in Breast Milk in Vaccinated Expectant Mothers
From Sept. 2022: Detection of Messenger RNA COVID-19 Vaccines in Human Breast Milk - PubMed (nih.gov). This study found some breastmilk samples contained mRNAs from COVID-19 shots. The safety of the mRNA-containing LNPs in infants is untested. Assurances come in the form of words (“tiny amount”). Spike-reactive T-cells from mothers as well as antibodies were found in the breastmilk of women vaccinated during pregnancy (Reference). No studies on the long-term rates of chronic illness in infants born to mothers who received mRNA shots during pregnancy have been conducted, and none are planned.
Spike-Induced Cell Fusion Makes Spike a Cellular Toxin
This paper describes the efficiency with which the SARS-CoV-2 spike protein leads to syncytia (the fusion of two or more human cells) and suggests that many of the side-effects of the vaccine (and COVID-19 ‘complications’) may be explained by the biotoxicity of the spike protein. The concerns are those we now know all too well: heart inflammation, neurological effects, and potentially, neoplasms.
This was published in December 2021. Another study was published confirming the predictions (See SARS-CoV-2 spike protein-mediated cardiomyocyte fusion may contribute to increased arrhythmic risk in COVID-19. The authors wrote: “SARS-CoV-2 spike protein-mediated cardiomyocyte fusion may contribute to increased arrhythmic risk in COVID-19”.
The FDA and CDC have not made any public warnings on the toxicity of the spike protein due to cell fusion.
Blindness
In one of the most clear-cut examples of pathogenic priming available, a new adverse outcome can be expected: Retinal occlusion. The risk is long-term and cumulative. Using analyses reminiscent of my study with Dr. Paul Thomas, the fact of the long-term cumulative risk is stunningly clear:
The authors wrote:
“The cumulative incidence of retinal vascular occlusion was significantly higher in the vaccinated cohort compared to the unvaccinated cohort, 2 years and 12 weeks after vaccination. The risk of retinal vascular occlusion significantly increased during the first 2 weeks after vaccination and persisted for 12 weeks. Additionally, individuals with first and second dose of BNT162b2 and mRNA-1273 had significantly increased risk of retinal vascular occlusion 2 years following vaccination, while no disparity was detected between brand and dose of vaccines.” (Reference)
I do not think we can expect any statement or warning from FDA or CDC on this new adverse event that is most likely caused by Pfizer and Moderna mRNA jabs.
CD8 T-Cell Dominant Hepatitis Following COVID-19 Vaccines
Numerous reports of autoimmune hepatitis following mRNA COVID-19 jab exposures have been published. Importantly, one such patient exhibited vaccine-specific immune cells in the liver. The authors found this by studying the intrahepatic infiltrate, which was enriched for CD8 T cells with SARS-CoV-2-specificity compared to the peripheral blood. Some of the example cases: Case 1, Case 2).
Autoimmune Depression
A recent meta-analysis (January 2023 found that studies with more than 50% of participants with a history of COVID-19 infection were found to be higher depression symptoms in the vaccinated compared to the unvaccinated. This study was motivated by earlier reports of psychiatric events occurring within 10 days of receipt of the mRNA jabs. The meta-analysis nevertheless somehow was entitled to mislead: “COVID-19 Vaccination Is Not Associated with Psychiatric Adverse Events: A Meta-Analysis - PMC (nih.gov)”
There are others, all consistent with Pathogenic Priming.
A commentary published in 2023 reported that:
“Data on adverse reactions during vaccination have been accumulated for these vaccines, and adverse events suspected of having an autoimmune mechanism, such as vaccine-induced immune thrombotic thrombocytopenia (VITT), autoimmune liver disease, Guillain-Barré syndrome, Graves’ disease, IgA nephropathy, rheumatoid arthritis, systemic lupus erythematosus, and adult-onset Still’s disease, have been reported.”
FDA and CDC Were Warned About Pathogenic Priming
It’s not as though this information is new. There is now a massive literature stemming from my original findings. IPAK was the first to report likely molecular mimicry from unsafe epitopes in SARS-CoV-2 proteins in April 2020.
This approach was validated in the lab by Vojdani et al.
In my next article, I will outline the primary reason why although these predictions were explicit and clear, and Pfizer, Moderna, FDA, and CDC had ample time to avoid the autoimmune epitopes in the SARS-CoV-2 spike protein, the findings we now see in the long-term real-world and retrospective studies were not found in the clinical trials.
Citations
Aldén M, Olofsson Falla F, Yang D, Barghouth M, Luan C, Rasmussen M, De Marinis Y. Intracellular Reverse Transcription of Pfizer BioNTech COVID-19 mRNA Vaccine BNT162b2 In Vitro in Human Liver Cell Line. Curr Issues Mol Biol. 2022 Feb 25;44(3):1115-1126. doi: 10.3390/cimb44030073. PMID: 35723296; PMCID: PMC8946961.
Boettler T, Csernalabics B, Salié H, Luxenburger H, Wischer L, Salimi Alizei E, Zoldan K, Krimmel L, Bronsert P, Schwabenland M, Prinz M, Mogler C, Neumann-Haefelin C, Thimme R, Hofmann M, Bengsch B. SARS-CoV-2 vaccination can elicit a CD8 T-cell dominant hepatitis. J Hepatol. 2022 Sep;77(3):653-659. doi: 10.1016/j.jhep.2022.03.040. Epub 2022 Apr 21. PMID: 35461912; PMCID: PMC9021033.
Clemens DJ, Ye D, Zhou W, Kim CSJ, Pease DR, Navaratnarajah CK, Barkhymer A, Tester DJ, Nelson TJ, Cattaneo R, Schneider JW, Ackerman MJ. SARS-CoV-2 spike protein-mediated cardiomyocyte fusion may contribute to increased arrhythmic risk in COVID-19. PLoS One. 2023 Mar 8;18(3):e0282151. doi: 10.1371/journal.pone.0282151. PMID: 36888581; PMCID: PMC9994677.
Doerfler W. Adenoviral Vector DNA- and SARS-CoV-2 mRNA-Based Covid-19 Vaccines: Possible Integration into the Human Genome - Are Adenoviral Genes Expressed in Vector-based Vaccines? Virus Res. 2021 Sep;302:198466. doi: 10.1016/j.virusres.2021.198466. Epub 2021 Jun 1. PMID: 34087261; PMCID: PMC8168329.
Hanna N, Heffes-Doon A, Lin X, Manzano De Mejia C, Botros B, Gurzenda E, Nayak A. Detection of Messenger RNA COVID-19 Vaccines in Human Breast Milk. JAMA Pediatr. 2022 Dec 1;176(12):1268-1270. doi: 10.1001/jamapediatrics.2022.3581. Erratum in: JAMA Pediatr. 2022 Nov 1;176(11):1154. PMID: 36156636; PMCID: PMC9513706.
Lazebnik Y. Cell fusion as a link between the SARS-CoV-2 spike protein, COVID-19 complications, and vaccine side effects. Oncotarget. 2021 Dec 7;12(25):2476-2488. doi: 10.18632/oncotarget.28088. PMID: 34917266; PMCID: PMC8664391.
Lee SE, Shim SR, Youn JH, Han HW. COVID-19 Vaccination Is Not Associated with Psychiatric Adverse Events: A Meta-Analysis. Vaccines (Basel). 2023 Jan 16;11(1):194. doi: 10.3390/vaccines11010194. PMID: 36680038; PMCID: PMC9861090.
Li JX, Wang YH, Bair H, Hsu SB, Chen C, Wei JC, Lin CJ. Risk assessment of retinal vascular occlusion after COVID-19 vaccination. NPJ Vaccines. 2023 May 2;8(1):64. doi: 10.1038/s41541-023-00661-7. PMID: 37130882; PMCID: PMC10153772.
Sfera A, Hazan S, Anton JJ, Sfera DO, Andronescu CV, Sasannia S, Rahman L, Kozlakidis Z. Psychotropic drugs interaction with the lipid nanoparticle of COVID-19 mRNA therapeutics. Front Pharmacol. 2022 Sep 9;13:995481. doi: 10.3389/fphar.2022.995481. PMID: 36160443; PMCID: PMC9503827.
Yoshimi R, Nakajima H. COVID-19 Vaccination and the Development of Autoimmune Diseases. Intern Med. 2023 May 15;62(10):1387-1388. doi: 10.2169/internalmedicine.1490-22. Epub 2023 Mar 8. PMID: 36889704.
Zhang L, Richards A, Barrasa MI, Hughes SH, Young RA, Jaenisch R. Reverse-transcribed SARS-CoV-2 RNA can integrate into the genome of cultured human cells and can be expressed in patient-derived tissues. Proc Natl Acad Sci U S A. 2021 May 25;118(21):e2105968118. doi: 10.1073/pnas.2105968118. PMID: 33958444; PMCID: PMC8166107.
University at Buffalo News Releases News Topics Media Advisories University Statements Big data study refutes anti-vax blood clot claims about COVID-19 vaccines https://www.buffalo.edu/news/releases/2023/04/015.html
Related:















The real science and risks were indisputable to anyone paying close attention. And since last year, thanks to the excellent in-depth legal research done by Katherine Watt at bailiwicknews.substack.com, we know why and how real science was usurped.
Long before it was known the possible shedding of the spike proteins from vaccinated people I abstain from visiting any of my families who recently got the vaccine first two doses and then there after the booster shots because in the past every time someone in the family got a flu shot especially the nasal one I always notice grandma got sick when the kids went visiting her I told the parents please abstain for a while after the vaccines flu shots before visiting any elderly person because they may have manifested the disease in themselves as a carrier again I’m not a scientist I had no proof of this this is just my feeling at the time , during the holidays everyone who was vaccinated they got together for Christmas and Thanksgiving got sick because of the grouping together and I know without a shadow of a doubt if I went visiting them during that time I would’ve been sick from them i’m not vaccinated and to date I haven’t been sick since the beginning of the pandemic , again total antidotal I know but since there’s so much miss information from our governments big Pharma I had I to go by what I observed in my own little world ……